|
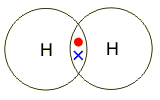
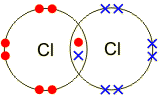
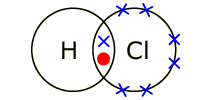
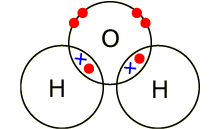
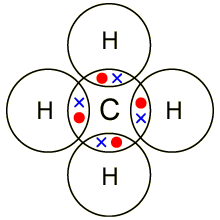
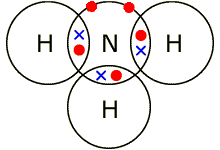
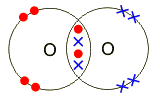
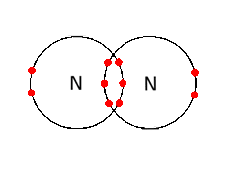
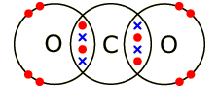
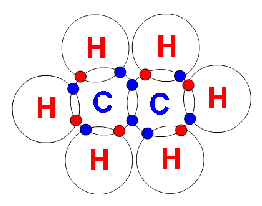
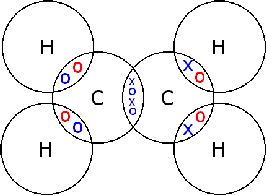
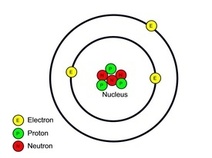
Electrons, being shared by atoms in a covalent bond, are attracted to the nucleus of each atom in the bond.
Electrons - Negatively Charged, Protons - Positively Charged A covalent bond forms when two NON-METAL atoms SHARE a pair of electrons.
(e.g. if one atom needs to gain 3 electrons, it will share with an atom that needs to lose 3 electrons)
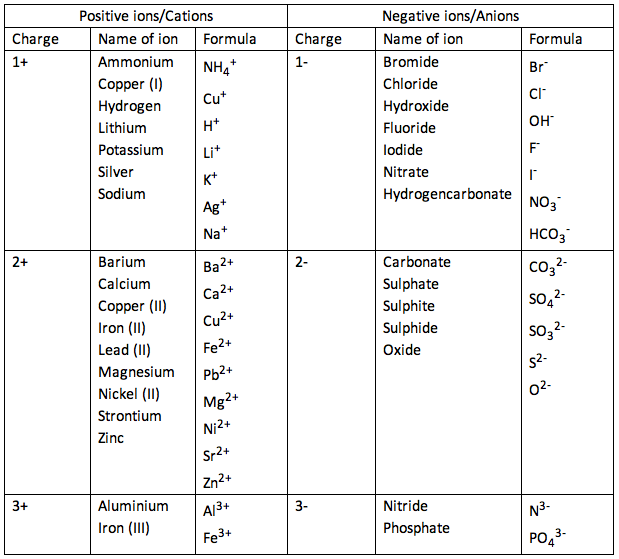
Electronic configuration of ions
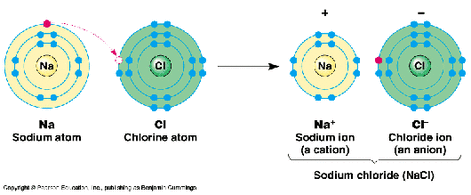
Na has the electronic configuration of 2,8,1 It's positive ion, Na+, has the electronic configuration of 2,8 Na loses an electron to another atom, therefore becomes positive. Cl has the electronic configuration of 2,8,7 It's negative ions, Cl-, has the electronic configuration of 2,8,8 Cl gains an electron from another atom, therefore becomes negative. 1.28 Understand oxidation as the loss of electrons and reduction as the gain of electrons11/21/2013 OILRIG
Oxidation Is Loss Reduction Is Gain Loss of an electron - Positively Charged Ions
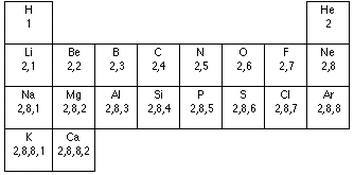
Gain of an electron - Negatively Charged Ions Group 1 - 3 will form CATIONS (Postive Ions) Group 5 - 7 will form ANIONS (Negative Ions) Group 0 & 8 are unreactive, therefore they do not form ions. The group number of the element represents the number of electrons on the elements outer most shell. The periods represent the number of shells/orbitals the element has.
1.12 Understand that the periodic table is an arrangement of elements in order of atomic number11/18/2013 Elements in the periodic table are arranged by atomic number. The number of protons in the element's atom increases across the periodic table. Columns are called groups, whilst rows are calleds periods. Metals occur on the left side of the table, as the non-metals occur on the right side of the table.
1.11 Calculate the relative atomic mass of an element from the relative abundances of its isotopes.11/18/2013 For example, the most common isotope of chlorine is 35, which accounts for around 75% of the atoms in chlorine. However, the isotope 37 accounts for arround 25% of all chlorine atoms. Therefore, the average atomic mass of all chlorine isotopes is:
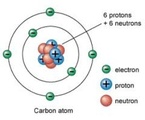
(75+35) + (25+37) ---------------------- = 35.5 100 However, there is no isotope of chlorine with 35.5 nucleons, so the RAM of Chlorine is 35. 1.10 Understand the terms atomic number, mass number, isotopes and relative atomic mass (Ar)11/18/2013 Atomic Number - The total number of protons in an atom.
Mass Number - The total number of sub-atomic particles inside the nucleus. Isotopes - These are atoms of the same element with the same number of protons but different number of neutrons. Relative Atomic Mass (Ar) - This is the combined relative masses of the nucleons in one particular isotope or an average of several isotopes. 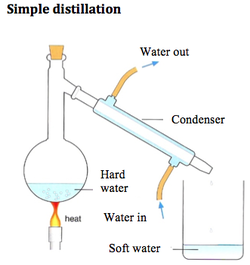 FOR THIS TOPIC, YOU ONLY NEED TO KNOW SIMPLE DISTILLATION. Simple Distillation - To obtain a solvent from a solution, simple distillation can be used. The solution is heated, the liquid boils and turns into vapour, and the vapour is condensed into the pure liquid to be collected. The impurities are left behind, as they have far greater boiling points. Elements - Are molecules or atoms of only one type of atom
Mixtures - Consists of several different elements that have not undergone a chemical reaction Compounds - Consists of several different elements, which have undergone a chemical reaction and bonded  DILUTION 1. Place a crystal of potassium manganate(VII) in a beaker of water. 2. The color spreads through the water. 3. Over time you can see the color levels out but it is weaker than the original colour. This is because particles leave the crystal, it dissolves. Then they mix among the water particles. 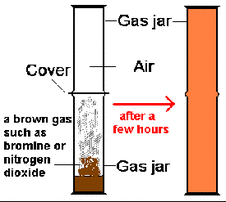 DIFFUSION 1. Place an open gas jar of air upside down on an open gas jar containing a few drops of red-brown bromine. 2. The color spreads upwards This is because the particles of bromine vapour mix among the particles of air. SOLID: STATIONARY; NO ENERGY; ALL PARTICLES TOUCHING/COMPACT
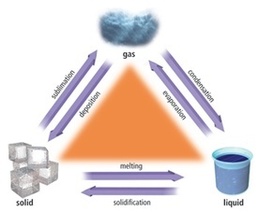
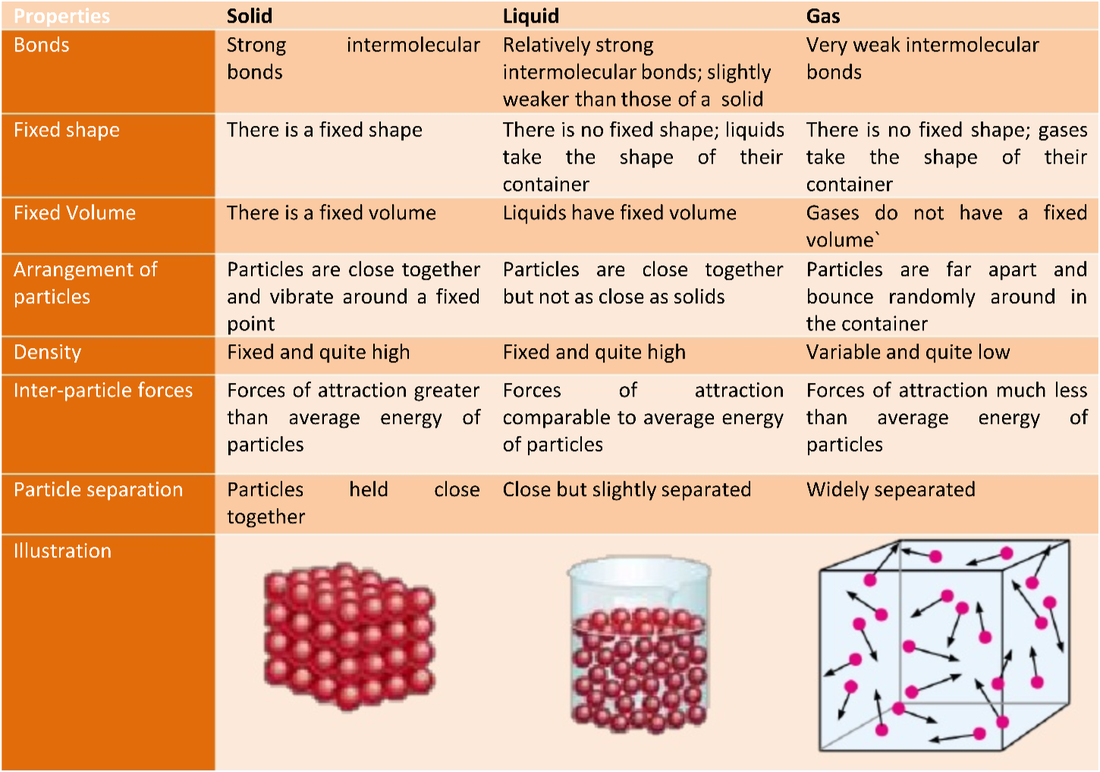
MELTING LIQUID: MOBILE TO FILL SPACE; SOME ENERGY; ALL PARTICLES TOUCHING/LESS COMPACT BOILING GAS: MOVING FREELY; LOTS OF ENERGY; PARTICLES NOT TOUCHING/LOOSE |