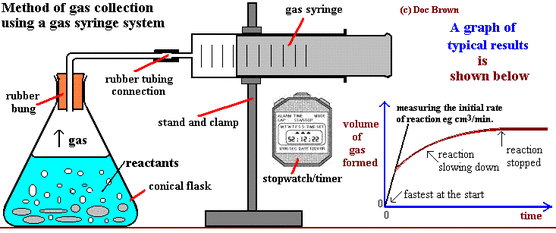
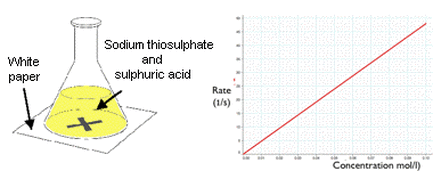
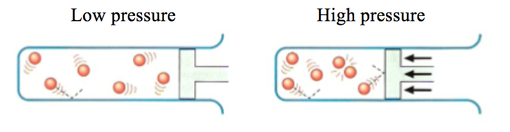
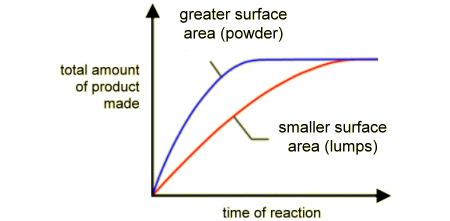
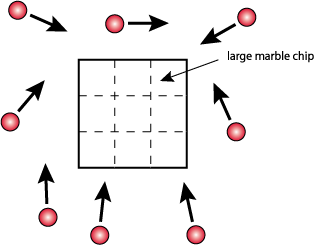
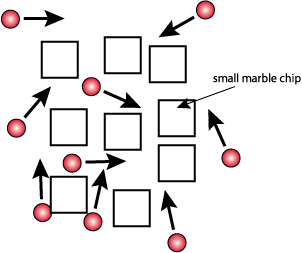
Surface Area -
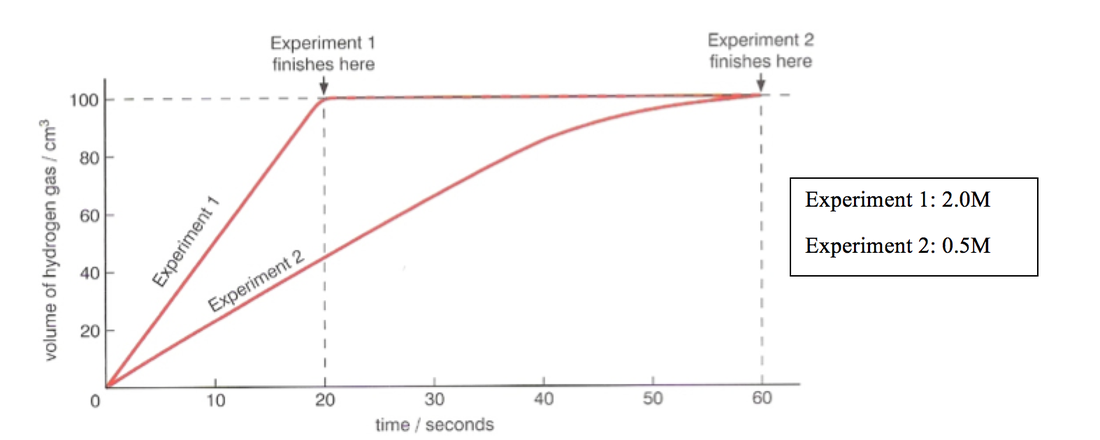
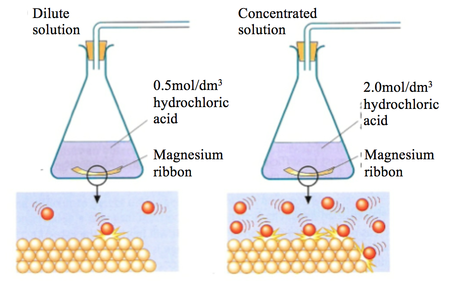
- Obtain 4 large marble chips (CaCO3) of approximately the same size
- Measure 25cm3 of HCl into a conical flask with a gas syringe attached
- Crush 2 marble chips into small pieces using a mortar and pestle
- Add this to the acid and record the volume of CO2 at 20 second intervals until it reaches 100cm3 of HCl
- Use mortar and pestle to crush other 2 chips into a powder
- Repeat step 4
Concentration -
- Measure 50cm3 of Sodium Thiosulphate Solution (Na2S2C3) into a conical flask
- Measure 5cm3 of HCl
- Draw a large, bold 'X' and place conical flask on top of it
- Add HCl to Sodium Thiosulphate solution and start stopwatch.
- Cover the flask with cling film
- A yellow solution will form
- When you can no longer see the 'X', stop the stopwatch and record the time
- Repeat the experiment again with a different volume of Thiosulphate.
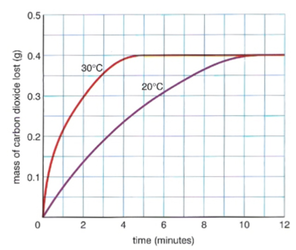
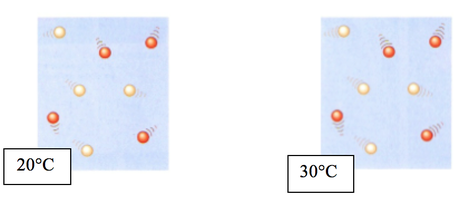
Temperature -
- Put 25cm3 of HCl in a small conical flask using a measuring cylinder
- Use a tripod and a bunsen burner to heat the acid to the required temperature range
- *DO NOT HEAT ACID ABOVE 70 DEGREES*
- Take an accurate temperature with a thermometer
- Cut a piece of magnesium strip into 1 cm
- Put the magnesium strip into the acid and cover it with a cotton wool
- Start a stopwatch and time until the magnesium has dissolved
- Repeat this with another temperature range