To measure the effect of concentration on the rate of reaction, a different experiment can be done.
A piece of magnesium ribbon can be added to a solution of hydrochloric acid. The following reaction occurs:
Magnesium + hydrochloric acid --> magnesium chloride + hydrogen
Two experiments could be performed using different concentrations of acid (say 2.0mol/l and 0.5mol/l).
Result
Pressure
A piece of magnesium ribbon can be added to a solution of hydrochloric acid. The following reaction occurs:
Magnesium + hydrochloric acid --> magnesium chloride + hydrogen
Two experiments could be performed using different concentrations of acid (say 2.0mol/l and 0.5mol/l).
Result
- The test tube containing the higher concentration of hydrochloric acid should react faster and produce more hydrogen faster than the other test tube
- The higher the concentration, the more frequent the collision
- The more collisions, the rate of reaction increases
- Therefore, increasing concentration increases the rate of reaction
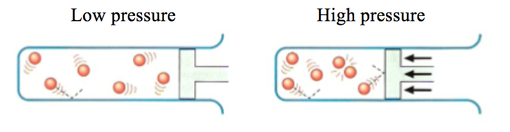
Pressure
- Increasing the pressure on a reaction between gases will increase the rate of reaction.
- Increasing the pressure reduces the volume of gas, moving the particles closer together.
- If the particles are denser, there will be more collisions per second and therefore more effective collisions per second.