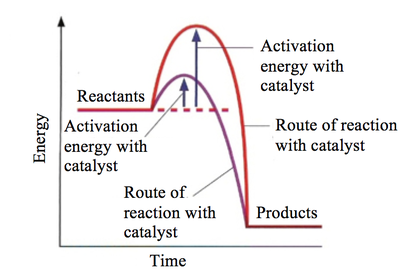
A catalyst is a substance that speeds up the rate of a chemical reaction without being used up itself. The mass of the catalyst remains unchanged for the whole reaction. It provides an alternative pathway and lowers the activation energy
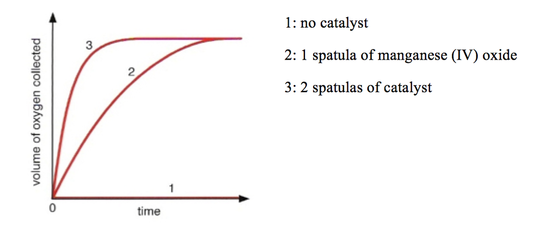
To explore the effects of a catalyst, there is a very effective experiment that could be done:
Hydrogen peroxide → water + oxygen
This reaction would take a very long time to occur, but by adding a catalyst (in this case manganese (IV) oxide), it will make the reaction occur quicker. This demonstrates that the presence of a catalyst increases the rate of reaction.
To explore the effects of a catalyst, there is a very effective experiment that could be done:
Hydrogen peroxide → water + oxygen
This reaction would take a very long time to occur, but by adding a catalyst (in this case manganese (IV) oxide), it will make the reaction occur quicker. This demonstrates that the presence of a catalyst increases the rate of reaction.
Most catalysts work by providing an alternative route for the reaction, lowering the activation energy. This increases the number of collisions, hence increases the rate of reaction.