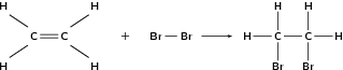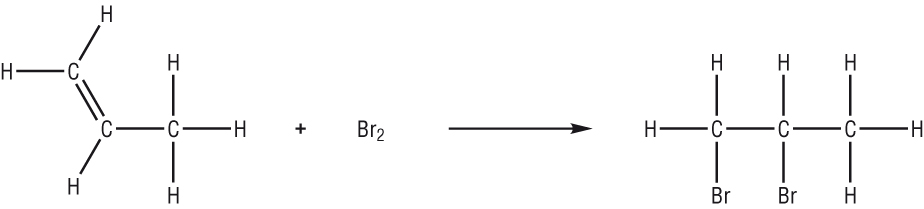|
4 Comments
Conditions
CH4 + BR2 --> CH3Br + HBr
Methane + Bromine --> Bromomethane + Hydrogen Bromide (UV Light) A substitution reaction occurs because hydrogen is replaced with bromine General Formula: CnH2n
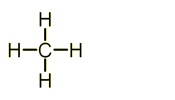
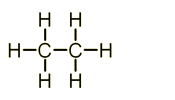
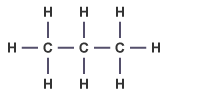
General Formula = Cn + H2n+2
Homologous Series is a series of chemical compounds having the same functional group but differing in composition by a fixed group of atoms. The compounds have the following in common:
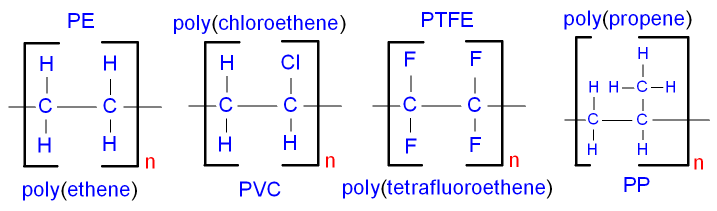
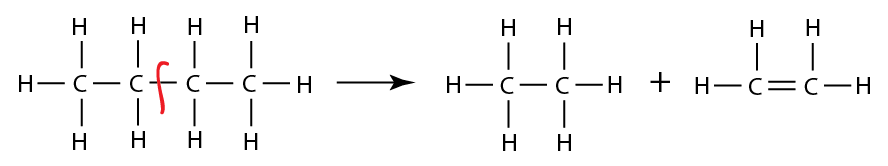
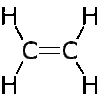
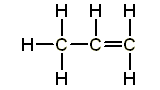
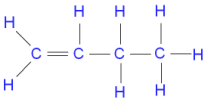
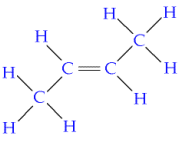
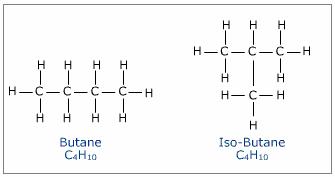
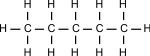
Saturated contain only single C-C bonds (Alkanes) Unsaturated contain a double C=C bond (Alkenes) General Formula Organic compounds in the same homologous series all have the same general formula (eg. Alkanes - CnH2n+2) Isomerism is the existence of two or more compounds with the same molecular formula but different structural formula (eg. Butane) Delta H = molar enthalpy change
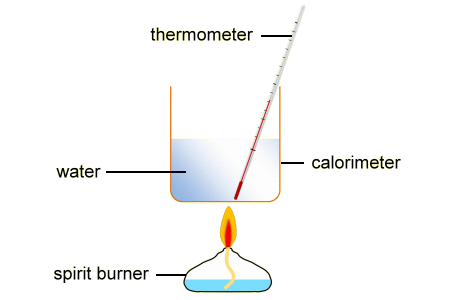
Specific heat capacity = 4.2 J/g Mass of water = mass of water in grams Delta t = Temperature increase/decrease by the reaction
2.24 understand that carbon dioxide is a greenhouse gas and may contribute to climate change11/23/2013
Incomplete combustion of hydrocarbons produce
Combustion
Alkane + Oxygen --> Carbon Dioxide + Water Incomplete Combustion Alkane + Little Oxygen --> Carbon Monoxide + Water
Hydrocarbons = organic compound that contains hydrogen & carbon atoms only.
|