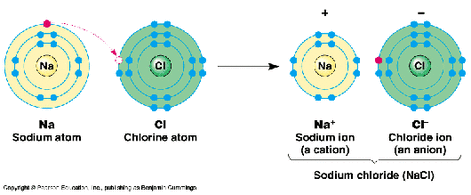
- An electronic transfer takes place
- Na has one electron on it's outer shell, therefore it wants to lose it to have a complete outer shell
- Na gives one electron to Cl
- Cl has seven electrons on it's outer shell, therefore it wants to gain one electron to have a complete outer shell
- Cl gains one electron from N
- The Na atom becomes a positive ion by losing one electron
- The Cl atom becomes a negative ion by gaining one electron