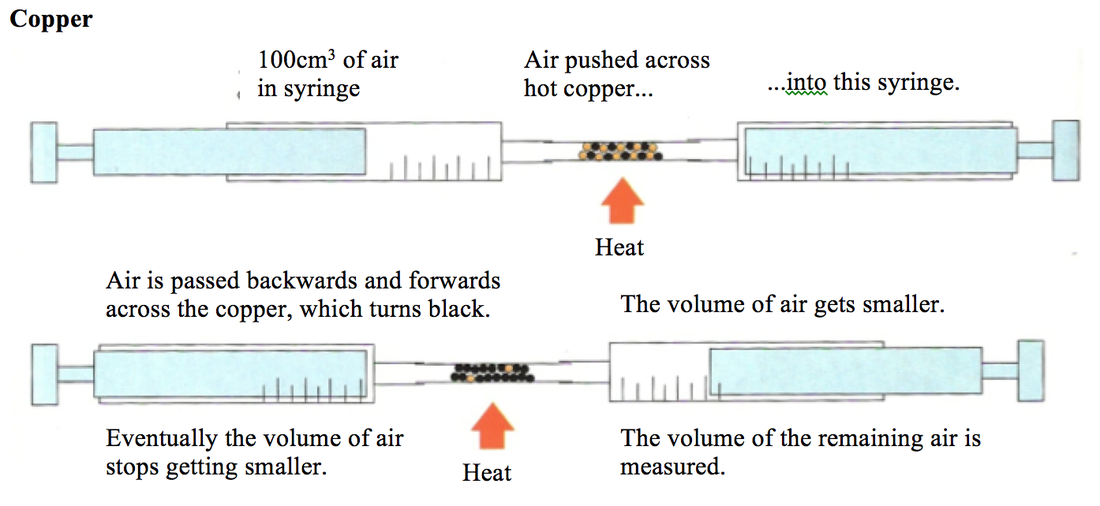
The copper is in excess and uses up the oxygen to form copper oxide (CuO). All the oxygen in the air is therefore used up, and so the volume of the air decreases by about 20% (the percentage of oxygen in air).
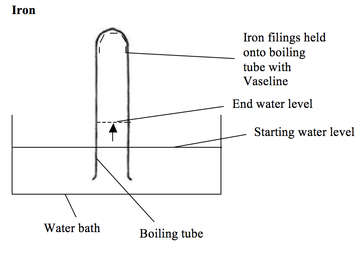
The iron reacts with the oxygen in the air (it rusts). The water level therefore rises by about a fifth over several days.
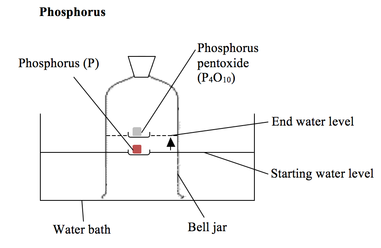
The phosphorus is lit with a hot wire. It reacts with the oxygen in the air and causes the water level in the bell jar to rise by about 20%.