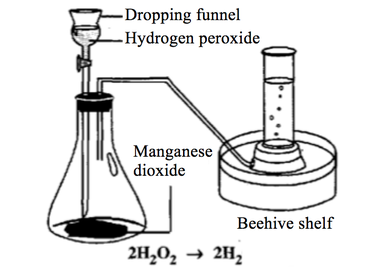
Hydrogen Peroxide --> Water + Oxygen
H2O2(aq) --> H2O(l) + O2(g)
The rate of decomposition is very slow without a catalyst. However, when manganese (IV) oxide powder is added to the hydrogen peroxide solution, oxygen is produced rapidly.
H2O2(aq) --> H2O(l) + O2(g)
The rate of decomposition is very slow without a catalyst. However, when manganese (IV) oxide powder is added to the hydrogen peroxide solution, oxygen is produced rapidly.